Whether you are a software developer, an engineer, or a R&D manager designing a medical device, software quality is critical for patient safety.
Model-Based Design incorporates verification and validation into the software development workflow. As a result, the software is comprehensively tested and verified before you integrate it into a medical device. In addition, most of the documentation required by the IEC 62304 standard are automatically generated for regulatory compliance.
But how does Model-Based Design work in practice? And how do you get started?
Learn more with these white papers.
Model-Based Design for Medical Device Development
Learn about Model-Based Design with real-world examples that help teams reduce development time, minimize integration issues, and deliver safety-critical products while complying with regulations and standards.
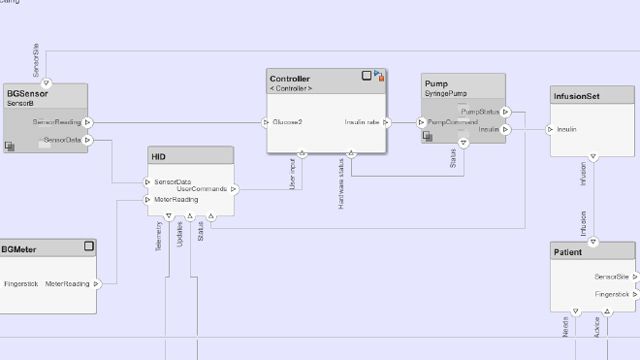
Best Practices for IEC 62304 Compliance with Model-Based Design
Learn how Model-Based Design with MATLAB and Simulink can be used to develop medical devices in higher severity safety class levels B and C, following the main activities identified in the IEC 62304 standard.

